Abstract
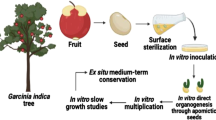
Ensets (Ensete spp., family Musaceae), are multipurpose plants with great versatility and potential as a food security crop, although they are cultivated only in Ethiopia and few other regions of Africa. Currently due to loss of habitat, intensification of agriculture, genetic diversity of both wild and domestic ensets is at high risk of erosion. Ensete glaucum (Roxb.) Cheesman, a less-known and under-utilized crop wild relative of banana, is found as natural strands in North-eastern regions of India. No report exists on seed conservation or cryopreservation of E. glaucum and the present work was undertaken to fill this knowledge gap. Mature fruits of two seeded accessions of E. glaucum (MZU-DBT-T15 and HSB-8) were collected from plants in natural populations from two geographically distinct North-eastern region of India. Seed morphology, moisture content (MC) of seed and embryo, viability and germination of excised embryos, desiccation and cryosensitivity were studied. Fresh seed (31% MC) viability measured through in vitro germination of excised embryos was not significantly different in both accessions (87–89%). Seeds could tolerate desiccation up to 5–10% MC without significant loss in germination potential (83–84%) and safely stored up to 1 year at room temperature (RT), without loss of viability. Air dehydrated seeds (10 ± 2% MC) and embryos (12 ± 2% MC) were cryopreserved (in liquid and vapour phase of liquid nitrogen). High post-thaw recovery was achieved in both tissues i.e. seeds (80.2 ± 2.2%) and embryos (83.1 ± 2.6%) by in vitro culture of excised embryos on MS medium + 2 µM gibberellic acid + 1 µM 6-benzyl amino purine (BAP) + 1 µM ascorbic acid. The seedlings converted into multiple shoots (15–20 shoots/explant) within 2 months of culture on medium with MS salts + 10 µM BAP + 1 µM indole-3-acetic acid (IAA) + 1 µM ascorbic acid. This study is the first successful report of cryopreservation of E. glaucum using simple air dehydration. Using the method devised, seeds and zygotic embryos of the two accessions of E. glaucum are safely conserved in the Cryogenebank, while in vitro plantlets are maintained in the In Vitro Genebank of ICAR-NBPGR, New Delhi, India.
Key message
This study is the first successful report of cryopreservation of Ensete glaucum using seeds and zygotic embryos





Similar content being viewed by others
Abbreviations
- MC:
-
Moisture content
- DAF:
-
Days after flowering
- BAP:
-
6-Benzyl amino purine
- IAA:
-
Indole-3-acetic acid
- GA3 :
-
Gibberellic acid
- MS:
-
Murashige and Skoog medium
- LN:
-
Liquid nitrogen
- RT:
-
Room temperature
References
Abdelnour-Esquivel A, Mora A, Villalobos V (1992) Cryopreservation of zygotic embryos of Musa acuminata (AA) and Musa balbisiana (BB). CryoLetters 13:159–164
Afele JC, De Langhe E (1991) Increasing in vitro germination of Musa balbisiana seed. Plant Cell Tissue Organ Cult 27(1):33–36. https://doi.org/10.1007/BF00048203
Agrawal A, Swennen R, Panis B (2004) A comparison of four methods for cryopreservation of meristems in banana (Musa spp.). CryoLetters 25:101–110
Agrawal A, Tyagi RK, Goswami R (2008) Cryopreservation of subgroup Monthan (ABB) of Indian cooking bananas (Musa spp.). Curr Sci 94:1125–1128
Bezuneh T (1971) The germination and seedling development of Ensete SSP. East Afr Agric For J 37(2):156–159. https://doi.org/10.1080/00128325.1971.11662518
Bezuneh T (1980) The morphology of Ensete embryo in vitro culture. Eth J Agric Sci 2:23–28
Bhat SR, Bhat KV, Chandel KPS (1994) Studies on germination and cryopreservation of Musa balbisiana seed. Seed Sci Technol 22:637–640
Birmeta G, Welander M (2004) Efficient micropropagation of Ensete ventricosum applying meristem wounding: a three-step protocol. Plant Cell Rep 23:277–283. https://doi.org/10.1007/s00299-004-0832-9
Borrell JS, Biswas MK, Goodwin M, Blomme G, Schwarzacher T, Heslop-Harrison JS, Wendawek AM, Berhanu A, Kallow S, Janssens S, Molla EL (2019) Enset in Ethiopia: a poorly characterized but resilient starch staple. Ann Bot 123(5):747–766. https://doi.org/10.1093/aob/mcy214
Chin HF (1996) Germination and storage of banana seeds. In: Frison EA, Horry JP, De Waele D (eds) Proceedings of the workshop on new frontiers in resistance breeding for Nematodes, Fusarium and Sigatoka, Kuala Lumpur, pp 218–227
Diro M, van Staden J (2003) In vitro regeneration of Ensete ventricosum from zygotic embryos of stored seeds. S Afr J Bot 69(3):364–369. https://doi.org/10.1016/S0254-6299(15)30319-7
Diro M, van Staden J (2004a) Germination of zygotic embryos and in vitro growth of seedlings of wild types of Ensete ventricosum. S Afr J Bot 70(4):635–639. https://doi.org/10.1016/S0254-6299(15)30203-9
Diro M, van Staden J (2004b) Propagation of Ensete in vitro: a review. S Afr J Bot 70(4):497–501. https://doi.org/10.1016/S0254-6299(15)30187-3
Ellis RH, Hong TD, Roberts EH (1985) Handbook of seed technology for genebanks. Volume I. Principles and methodology. Handbooks for Genebanks, No. 2, International Board for Plant Genetic Resources, Italy
Gezahegn G, Mekbib F (2016) In vitro regeneration of disease free enset [Ensete ventricosum (Welw) Cheesman] planting materials from bacterial wilt diseased plants using shoot tip culture. Afr J Biotechnol 15(40):2192–2201
Graven P, De Koster CG, Boon JJ, Bouman F (1996) Structure and macromolecular composition of the seed coat of the Musaceae. Ann Bot 77(2):105–122. https://doi.org/10.1006/anbo.1996.0013
Guzzon F, Müller JV (2016) Current availability of seed material of enset (Ensete ventricosum, Musaceae) and its Sub-Saharan wild relatives. Genet Res Crop Evol 63(2):185–191. https://doi.org/10.1007/s10722-015-0355-y
ISTA (2016) International seed testing association RULES. ISTA Germination Sec., Chapter 19, pp 19–41
Joe A, Sreejith E, Sabu M (2016) Genus Ensete (Musaceae) in India. Telopea 19:99–112
Karlsson LM, Tamado T, Dalbato AL, Mikias Y (2013) Seed morphology and germination of Ensete ventricosum (Musaceae). Seed Sci Technol 41(3):357–370. https://doi.org/10.15258/sst.2013.41.3.04
Kulkarni VM, Varshney LR, Bapat VA, Rao PS (2002) Somatic embryogenesis and plant regeneration in a seeded banana (Ensete superbum (Roxb.) Cheesman). Curr Sci 83(8):939–941
Kumar VS, Jaishanker R, Annamalai A (2019) Variation studies in surface micromorphology on seed coat and endosperm of Ensete superbum (Roxb.) Cheesman: a conservation concern species of India. Curr Sci 117(9):1531–1536
Majumdar K, Sarkar A, Deb D, Majumder J, Datta BK (2013) Distribution record of Ensete glaucum (Roxb.) Cheesm. (Musaceae) in Tripura, Northeast India: a rare wild primitive banana. Asian J Cons Biol 2:164–167
Matheka J, Tripathi JN, Merga I, Gebre E, Tripathi L (2019) A simple and rapid protocol for the genetic transformation of Ensete ventricosum. Plant Methods 15:130. https://doi.org/10.1186/s13007-019-0512-y
Mathew MM, Philip VJ (1996) Clonal propagation of Enset (Ensete superbum (Roxb.) Cheesman) through shoot tip culture. Plant Cell Rep 16:232–234. https://doi.org/10.1007/BF01890874
Mathew MM, Philip VJ (2003) Somatic embryogenesis versus zygotic embryogenesis in Ensete superbum. Plant Cell Tissue Organ Cult 72:67–275. https://doi.org/10.1023/A:1022311126043
McGahan MW (1961) Studies on the seed of banana II. The anatomy and morphology of the seedling of Musa balbisiana. Am J Bot 48(7):630–637. https://doi.org/10.1002/j.1537-2197.1961.tb11690.x
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
MusaNet (2016) Global strategy for the conservation and use of Musa genetic resources (B. Laliberté, compiler). Bioversity International, Montpellier
Negash A, Puite K, Schaart J, Visser B, Krens F (2000) In vitro regeneration and micropropagation of enset from Southwestern Ethiopia. Plant Cell Tissue Organ Cult 62(2):153–158. https://doi.org/10.1023/A:1026701419739
Negash A, Krens F, Schaart J, Visser B (2001) In vitro conservation of enset under slow-growth conditions. Plant Cell Tissue Organ Cult 66(2):107–111. https://doi.org/10.1023/A:1010647905508
Normah MN, Sulong N, Reed BM (2019) Cryopreservation of shoot tips of recalcitrant and tropical species: advances and strategies. Cryobiology 87:1–14. https://doi.org/10.1016/j.cryobiol.2019.01.008
Nuraga GW, Feyissa T, Tesfaye K, Demissew S, Tadele Z (2019) Phenotypic diversity of enset (Ensete ventricosum (Welw.) Cheesman) landraces used in traditional medicine. Genet Resour Crop Evol 66:1761–1772. https://doi.org/10.1007/s10722-019-00832-3
Ochiai Y (2012) From forests to homegardens: a case study of Ensete glaucum in Myanmar and Laos. Tropics 21(2):59–66
Olango TM, Tesfaye B, Catellani M, Pè ME (2014) Indigenous knowledge, use and on-farm management of enset (Ensete ventricosum (Welw.) Cheesman) diversity in Wolaita, Southern Ethiopia. J Ethnobiol Ethnomed 10:41. https://doi.org/10.1186/1746-4269-10-41
Panis B (2009) Cryopreservation of Musa germplasm. In: Engelmann F, Benson E (eds) Technical Guidelines 9. Bioversity International, Montpellier
Panis B (2019) Sixty years of plant cryopreservation: from freezing hardy mulberry twigs to establishing reference crop collections for future generations. Acta Hortic 1234:1–7. https://doi.org/10.17660/ActaHortic.2019.1234.1
Panis B, Withers LA, De Langhe E (1990) Cryopreservation of Musa suspension cultures and subsequent regeneration of plants. CryoLetters 11:337–350
Panis B, Totte N, Van Nimmen K, Withers LA, Swennen R (1996) Cryopreservation of banana (Musa spp.) meristem cultures after preculture on sucrose. Plant Sci 121(1):95–106. https://doi.org/10.1016/S0168-9452(96)04507-4
Panis B, Strosse H, Van Den Hende S, Swennen R (2002) Sucrose preculture to simplify cryopreservation of banana meristem cultures. CryoLetters 23(6):375–384
Panis B, Piette B, Swennen R (2005) Droplet vitrification of apical meristems: a cryopreservation protocol applicable to all Musaceae. Plant Sci 168(1):45–55. https://doi.org/10.1016/j.plantsci.2004.07.022
Ponni TG, Nair AS (2019a) Somatic embryogenesis to overcome low seed viability and conserve wild banana (Ensete superbum (Roxb.) Cheesman). Vitro Cell Dev Biol Plant 55:371–379. https://doi.org/10.1007/s11627-019-09998-y
Ponni TG, Nair AS (2019b) In vitro cormlet production-an efficient means for conservation in Ensete superbum (Roxb.) Cheesman. J App Hortic 21:20–24
Rao AS, Hajra PK (1976) Ensete glaucum (Roxb.) Cheesman in the Khasi Hills, Meghalaya, India. Nelumbo 18(1–4):207–210
Roberts EH (1973) Predicting the storage life of seeds. Seed Sci Technol 1:499–514
Tesfaye M (1992) The effect of soaking, temperature and other pretreatments on the germination of enset’seed. Seed Sci Technol 20(3):533–538
Thinh NT (1999) Cryopreservation of in vitro-grown shoot tips of banana (Musa spp.) by vitrification method. Cryo-Letters 20:163–174
Tripathi J, Matheka J, Merga I, Gebre E, Tripathi L (2017) Efficient regeneration system for rapid multiplication of clean planting material of Ensete ventricosum (Welw.) Cheesman. Vitro Cell Dev Biol Plant 53:624–630. https://doi.org/10.1007/s11627-017-9867-9
Uma S (2006) Farmer’s knowledge of Wild Musa in India. Report FAO Rome and National Research Centre for Banana, Trichy
Vineesh PS, Skaria R, Mukunthakumar S, Padmesh P, Decruse SW (2015) Seed germination and cryostorage of Musa acuminata subsp. burmannica from Western Ghats. S Afr J Bot 100:158–163. https://doi.org/10.1016/j.sajb.2015.05.024
Wang MR, Lambardi M, Engelmann F, Pathirana R, Panis B, Volk GM, Wang QC (2020) Advances in cryopreservation of in vitro-derived propagules: technologies and explant sources. Plant Cell Tissue Organ Cult. https://doi.org/10.1007/s11240-020-01770-0
Yemataw Z, Chala A, Ambachew D, Studholme DJ, Grant MR, Tesfaye K (2017) Morphological variation and inter-relationships of quantitative traits in enset (Ensete ventricosum (welw.) Cheesman) germplasm from South and South-Western Ethiopia. Plants 6(4):56. https://doi.org/10.3390/plants6040056
Acknowledgements
Thanks are due to the Indian Council of Agricultural Research (ICAR) and Director, ICAR-NBPGR, New Delhi, for providing all the necessary laboratory facilities under an in-house project, during this research work. The financial support from the Department of Biotechnology, Ministry of Science and Technology, Government of India, New Delhi, under the Twining Scheme (Order No. BT/PR16128/NER/95/442/2015) is also sincerely acknowledged. We are thankful to the Editors and Reviewers for their constructive suggestions to improve the manuscript.
Author information
Authors and Affiliations
Contributions
AA conceived the idea, designed experiments, and wrote the manuscript. TR and GDH collected germplasm and provided support in project work. SS performed experiments, analyzed results and contributed to writing the manuscript. HS and DPSM provided the technical assistance in experiments. RK contributed in providing constructive feedback. All authors have reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Bart Panis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, S., Thangjam, R., Harish, G.D. et al. Conservation protocols for Ensete glaucum, a crop wild relative of banana, using plant tissue culture and cryopreservation techniques on seeds and zygotic embryos. Plant Cell Tiss Organ Cult 144, 195–209 (2021). https://doi.org/10.1007/s11240-020-01881-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01881-8