Abstract
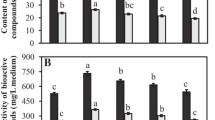
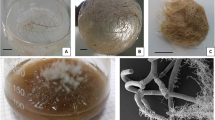
Gentiana species belonging to the Gentianaceae family are medicinal plants rich in glycosides and phenolics. Gentiana lutea L. is a highly sought-after plant for medicinal and industrial purposes because of its content of secondary metabolites. Not only G. lutea, but also Gentiana boissieri Schott et Kotschy ex Boiss., another Gentiana species distributed only in Turkey, is on the endangered species list and is not allowed to be collected in more than 100 countries. For this reason, the development of an efficient in vitro culture method to obtain the secondary metabolites synthesized in its roots is important, as it allows the production of higher amounts of these valuable metabolites. This study was conducted to determine the appropriate inoculum density (ID, fresh root weight per unit volume to be used), which is an important factor for biomass and secondary metabolite production, for root growth and production of phenolics and glycosides in the root culture of G. lutea and G. boissieri. For this purpose, roots were cultured at different IDs (5, 10, 15, 20, 25, and 30 g L−1) and evaluated for fresh root weight, root growth index, root dry weight, and content of iridoid (loganic acid), secoiridoids (swertiamarin, sweroside, gentinopicroside), xanthan (isogentisin), total phenolic, and some important phenolic compounds. All parameters were found to vary depending on the IDs. The result of the study was that the highest values in both root growth and production of most metabolites were obtained from the roots of G. lutea and G. boissieri cultivated with 20 g L−1 and 5 g L−1 IDs, respectively.



Similar content being viewed by others
References
Aberham A, Schwaiger S, Stuppner H, Ganzera M (2007) Quantitative analysis of iridoids, secoiridoids, xanthones and xanthone glycosides in Gentiana lutea L. roots by RP-HPLC and LC–MS. J Pharm Biomed Anal 45:437–442
Adelberg J, Toler J (2004) Comparison of agar and an agitated, thin-film, liquid system for micropropagation of ornamental elephant ears. Hort Sci 39:1088–1092
Ahmed S, Hahn EJ, Paek KY (2008) Aeration volume and photosynthetic photon flux affect cell growth and secondary metabolite contents in bioreactor cultures of Morinda citrifolia. J Plant Biol 51:209–212
Baque MA, Shiragi MHK, Moh SH, Lee EJ, Paek KY (2013) Production of biomass and bioactive compounds by adventitious root suspension cultures of Morinda citrifolia (L.) in a liquid-phase airlift balloon-type bioreactor. In Vitro Cell Dev Biol - Plant 49:737–749
Boateng A (2018) Hepatoprotective properties of Gentiana spp: against non-alcoholic fatty liver disease (NAFLD) (Doctoral dissertation, University of Westminster)
Cafaro T, Carnicelli V, Caprioli G, Maggi F, Celenza G, Perilli M, Bozzi A, Amicosante G, Brisdelli F (2020) Anti-apoptotic and anti-inflammatory activity of Gentiana lutea root extract. Adv Trad Med 20:619–630
Catană R, Holobiuc I, Mitoi M (2017) The antioxidant properties of Gentiana lutea root cultures. AgroLife 6:51–57
Catorci A, Piermarteri K, Tardella FM (2014) Pedo-climatic and land use preferences of Gentiana lutea subsp. lutea in central Italy. Plant Ecol Evol 147:176–186
Cheruvathur MK, Thomas TD (2014) Effect of plant growth regulators and elicitors on rhinacanthin accumulation in hairy root cultures of Rhinacanthus nasutus (L.) Kurz. Plant Cell Tiss Org Cult 118:169–177
Citová I, Ganzera M, Stuppner H, Solich P (2008) Determination of gentisin, isogentisin, and amarogentin in Gentiana lutea L. by capillary electrophoresis. J Separat Sci 31:195–200
Cui XH, Chakrabarty D, Lee EJ, Paek KY (2010) Production of adventitious roots and secondary metabolites by Hypericum perforatum L. in a bioreactor. Bioresource Technol 101:4708–4716
Cvetković S, Nastasijević B, Mitić-Ćulafić D, Đukanović S, Tenji D, Knežević-Vukčević J, Nikolić B (2020) New insight into the antigenotoxic activity of Gentiana lutea extracts–protective effect against food borne mutagens. Mutat Res Genet Toxicol Environ Mutagen 858:503251
Danphitsanuparn P, Boonsnongcheep P, Boriboonkaset T, Chintapakorn Y, Prathanturarug S (2012) Effects of Agrobacterium rhizogenes strains and other parameters on production of isoflavonoids in hairy roots of Pueraria candollei Grah. ex Benth. var. candollei. Plant Cell Tiss Org Cult 111:315–322
Davis PH (1985) Flora of Turkey and The East Aegean Islands. In: Davis PH, Cullen J, Coode MJE (eds) Vol 1–9, Edinburgh University Press, Edinburgh.
Deepthi S, Satheeshkumar K (2017) Effects of major nutrients, growth regulators and inoculum size on enhanced growth and camptothecin production in adventitious root cultures of Ophiorrhiza mungos L. Biochem Engin J 117:198–209
Drobyk NM, Mel’nyk VM, Hrytsak LR, Kravets NB, Konvalyuk II, Twardovska MO, Kunakh VA (2018) Establishment and analysis of tissue and fast-growing normal root cultures of four Gentiana L. species, rare highland medicinal plants. Biopoly Cell 34:461–476
Ekim T, Koyuncu M, Vural M, Duman H, Aytaç Z, Adıgüzel N (2000) Türkiye Bitkileri Kırmızı Kitabı, Barışcan Ofset, Türkiye Tabiatını Koruma Derneği, Van Yüzüncü Yıl Üniversitesi, Ankara Türkiye
Erken S, Kaleci N (2010) Censiyan (Gentiana lutea subsp. symphyandra) tohumlarının kontrollü koşullar altında çimlenme özelliklerinin belirlenmesi. Bahçe 39:17–26
Erken S, Özzambak ME (2017) Effect of light, temperature and different pretreatments on seed germination of Gentiana boissieri Schott et Kotschy ex Boiss. (Gentianaceae) an endemic to Turkey. Pak J Bot 49:41–46
Gambang K (2013) Synergism effect between inoculum size and aggregate size on flavonoid production in Centella asiatica (L.) Urban (Pegaga) cell suspension cultures. Intl J Res Engineer Technol 2:244–253
Glatthaar B, Saalmüller A, Haunschild J, Amon A (2009) Antiviral activity of a composition of Gentiana lutea L., Primula veris L., Sambucus nigra L., Rumex spec and Verbena officinalis L. (Sinupret®) against viruses causing respiratory infections. Eur J Integrat Med 1:258
Göktürk Baydar N, Babalik Z, Hallaç Türk F, Çetin ES (2011) Phenolic composition and antioxidant activities of wines and extracts of some grape varieties grown in Turkey. J Agri Sci 17:67–76
Halder M, Roychowdhury D, Jha S (2018) A critical review on biotechnological interventions for production and yield enhancement of secondary metabolites in hairy root cultures. In: Srivastava V, Mehrotra S, Mishra S (eds) Hairy roots. Springer, Singapore, pp 21–44
Haraguchi H, Tanaka Y, Kabbash A, Fujioka T, Ishizu T, Yagi A (2004) Monoamine oxidase inhibitors from Gentiana lutea. Phytochemistry 65:2255–2260
Ho TT, Lee KJ, Lee JD, Bhushan S, Paek KY, Park SY (2017) Adventitious root culture of Polygonum multiflorum for phenolic compounds and its pilot-scale production in 500 L-tank. Plant Cell Tiss Org Cult 130:167–181
Holobiuc I, Blindu R (2008) In vitro culture of the protected rare species Gentiana lutea L. for conservative purpose. Contribut Bot 42:125–134
Jensen SR, Schripsema J (2002) Chemotaxonomy and pharmacology of Gentianaceae. Gentianaceae-systematics and natural history, vol 5. Cambridge University Press, Cambridge, pp 574–631
Jeong CS, Murthy HN, Hahn EJ, Lee HL, Paek KY (2009) Inoculum size and auxin concentration influence the growth of adventitious roots and accumulation of ginsenosides in suspension cultures of ginseng (Panax ginseng C.A. Meyer). Acta Physiol Plant 31:219–222
Jeong CS, Murthy HN, Hahn EJ, Paek KY (2008) Improved production of ginsenosides in suspension cultures of ginseng by medium replenishment strategy. J Biosci Bioengineer 105:288–291
Jiao J, Gai QY, Fu YJ, Ma W, Yao LP, Feng C, Xia XX (2015) Optimization of Astragalus membranaceus hairy roots induction and culture conditions for augmentation production of astragalosides. Plant Cell Tiss Org Cult 120:1117–1130
Joksic G, Radak D, Sudar-Milovanovic E, Obradovic M, Radovanovic J, Isenovic ER (2021) Effects of Gentiana lutea root on vascular diseases. Curr Vascul Pharmacol 19:359–369
Joksić G, Tričković JF, Joksić I (2019) Potential of Gentiana lutea for the treatment of obesity-associated diseases. Curr Pharm Des 25:2071–2076
Lee EJ, Moh SH, Paek KY (2011) Influence of inoculum density and aeration volume on biomass and bioactive compound production in bulb-type bubble bioreactor cultures of Eleutherococcus koreanum Nakai. Bioresource Technol 102:7165–7170
Lee CW, Shuler ML (2000) The effect of inoculum density and conditioned medium on the production of ajmalicine and catharanthine from immobilized Catharanthus roseus cells. Biotechnol Bioengineer 67:6–1–71
Lee KJ, Park Y, Kim JY, Jeong TK, Yun KS, Paek KY, Park SY (2015) Production of biomass and bioactive compounds from adventitious root cultures of Polygonum multiflorum using air-lift bioreactors. J Plant Biotechnol 42:34–42
Ling APK, Chin MF, Hussein S (2009) Adventitious root production of Centella asiatica in response to plant growth regulators and sucrose concentration. Med Aromat Plant Sci Biotechnol 3:36–41
Menkovik N, Savikin K, Cebedzik R (1999) Investigation of the activity of Gentiana lutea extracts against Mycobacterium bovis. Pharm Pharmacol Lett 9:74–75
Min JY, Jung HY, Kang SM, Kim YD, Kang YM, Park DJ, Prasad DT, Choi MS (2007) Production of tropane alkaloids by small-scale bubble column bioreactor cultures of Scoplia parviflora adventitious roots. Bioresource Technol 98:1748–1753
Mirzaee F, Hosseini A, Jouybari HB, Davoodi A, Azadbakht M (2017) Medicinal, biological and phytochemical properties of Gentiana species. J Trad Complement Med 7:400–408
Moreno PR, Schlatmann JE, van der Heijden R, van Gulik WM, ten Hoopen HJ, Verpoorte R, Heijnen JJ (1993) Induction of ajmalicine formation and related enzyme activities in Catharanthus roseus cells: effect of inoculum density. Appl Microbiol Biotechnol 39:42–47
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Murthy HN, Dalawai D, Bhat MA, Dandin VS, Paek KY, Park SY (2021) Biotechnological production of useful phytochemicals from adventitious root cultures. In: Ramawat KG, Ekiert HM, Goyal S (eds) Plant cell and tissue differentiation and secondary metabolites: fundamentals and applications. Springer, Cham, pp 469–485
Mustafa AM, Caprioli G, Ricciutelli M, Maggi F, Marín R, Vittori S, Sagratini G (2015) Comparative HPLC/ESI-MS and HPLC/DAD study of different populations of cultivated, wild and commercial Gentiana lutea L. Food Chem 174:426–433
Nagella P, Chung IM, Murthy HN (2011) In vitro production of gymnemic acid from cell suspension cultures of Gymnema sylvestre R. Br Engineer Life Sci 11:537–540
Pan Y, Zhao YL, Zhang J, Li WY, Wang YZ (2016) Phytochemistry and pharmacological activities of the genus Gentiana (Gentianaceae). Chem Biodivers 13:107–150
Petrova M, Zagorska N, Tasheva K, Evstatieva L (2006) In vitro propagation of Gentiana lutea L. Genet Breed 35:63–68
Praveen N, Murthy HN (2010) Production of withanolide-A from adventitious root cultures of Withania somnifera. Acta Physiol Plant 32:1017–1022
Pringle JS (2014) Morphological characteristics of the family Gentianaceae. In: Rybczyński J, Davey M, Mikuła A (eds) The Gentianaceae-vol 1: characterization and ecology. Springer, Berlin, pp 1–12
Rahmat E, Kang Y (2019) Adventitious root culture for secondary metabolite production in medicinal plants: a review. J Plant Biotechnol 46:143–157
Rossi M, Fisogni A, Nepi M, Quaranta M, Galloni M (2014) Bouncy versus idles: on the different role of pollinators in the generalist Gentiana lutea L. Flora 209:164–171
Saha P, Mandal S, Das A, Das S (2006) Amarogentin can reduce hyperproliferation by downregulation of Cox-II and upregulation of apoptosis in mouse skin carcinogenesis model. Cancer Lett 244:252–259
Sakurai M, Mori T, Seki M, Furusaki S (1996) Changes of anthocyanin composition by conditioned medium and cell inoculum size using strawberry suspension culture. Biotechnol Lett 18:1149–1154
Savikin K, Menković N, Zdunić G, Stević T, Radanović D, Janković T (2009) Antimicrobial activity of Gentiana lutea L. extracts. Z Naturfor C 64:339–342
Savran A, Bağcı Y, Onat TA (2015) Niğde İli Sınırları İçinde Yayılış Gösteren Bazı Lokal Endemik Bitkilerin Taksonomik ve Ekolojik Özellikleri. Selçuk Üniversitesi Fen Fakültesi Fen Dergisi 41:1–54
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Amer J Enol Viticult 16:144–158
Wu CH, Dewir YH, Hahn EJ, Paek KY (2006) Optimization of culturing conditions for the production of biomass and phenolics from adventitious roots of Echinacea angustifolia. J Plant Biol 49:193–199
Wu S, Ning Y, Zhao Y, Sun W, Thorimbert S, Dechoux L, Sollogoub M, Zhang Y (2017) Research progress of natural product gentiopicroside-a secoiridoid compound. Mini Rev Med Chem 17:62–77
Yang JL, Liu LL, Shi YP (2010) Phytochemicals and biological activities of Gentiana species. Nat Prod Comm 5:1934578X1000500432
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aras Aşcı, Ö., Demirci, T., Albayrak, İ. et al. Optimization of inoculum density to support root growth and secondary metabolite accumulation in root cultures of endangered Gentiana species: Gentiana lutea and Gentiana boissieri. In Vitro Cell.Dev.Biol.-Plant 58, 1090–1098 (2022). https://doi.org/10.1007/s11627-022-10305-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-022-10305-5